Neutrophils are specialized innate immune cells that act as the first responders to any sign of infection or disease. This requires them to exert metabolic energy at a rapid rate.
In new research published in the journal Frontiers in Immunology, researchers at the Morgridge Institute for Research used advanced metabolic imaging to watch neutrophils at the moment they perform their quick-changing act.
“These cells are so much fun, but really hard to work with — you need so much patience,” says Rupsa Datta, an associate scientist in the lab of Morgridge Investigator Melissa Skala and first author of the study.
Despite being abundant in human blood, neutrophils are very short-lived and difficult to grow in culture, making them understudied and poorly understood.
The Skala Lab uses a specialized imaging technique called optical metabolic imaging (OMI) that Datta thought would be a good fit to help study this sensitive cell type.

“I wanted to observe how neutrophil metabolism changes with activation and see if our optical technique was sensitive to those changes,” Datta says. “You have to work fast and do everything the same day, maximum six hours after isolation. I wouldn’t do metabolic imaging after that.”
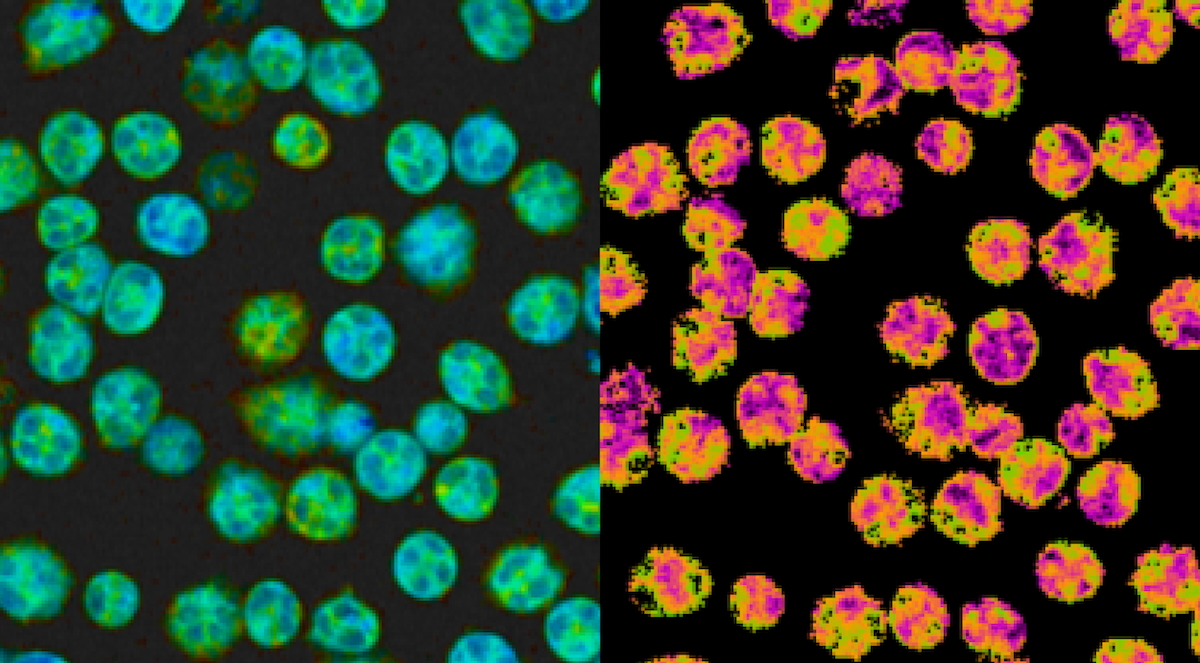
OMI is non-invasive and label-free by measuring the natural autofluorescence within cells instead of using chemical reagents that damage the cells. The method visualizes the rapid metabolic changes of cells in real-time and has the flexibility to be used with many different cell types in different systems.
Metabolic activity is measured by the reduction-oxidation state, or redox state, using a two-photon fluorescence lifetime microscopy (FLIM) system to measure the autofluorescence intensity and lifetimes of two metabolic enzymes, NAD(P)H and FAD. These signals are used together to calculate the optical redox ratio — a way to quantify the metabolic changes and shifts in energy production and consumption within a cell.
OMI also offers a way to gather real-time measurements from single cells, providing a unique look at the individual metabolic states of each cell within the neutrophil population.
The Jing Fan Lab at the Morgridge Institute collaborated with Datta to perform traditional metabolic assays such as mass spectrometry to characterize and compare metabolic data with OMI data.
“I also wanted to work with physiological activations for neutrophils,” Datta says. “Since they are the first responders to infection, I wanted to use actual pathogens.”
Datta sought the expertise of Gina Gallego-Lopez, also in the Skala Lab, who studies the parasite Toxoplasma gondii, with a collaboration rooted in curiosity.
“Toxoplasma is not the most typical pathogen that people would study with neutrophils,” she says. “But I remember sharing updates during a lab meeting and we both said, ‘Let’s just see what happens!’”
They also collaborated with Anna Huttenlocher, professor of medical microbiology & immunology at UW–Madison, who studies innate immune responses to infection, inflammation and disease. Her lab contributed work on neutrophils in response to the bacteria Pseudomonas aeruginosa, a common activator of neutrophils.
Together, these in vitro studies demonstrated that OMI was sensitive to detecting metabolic changes in neutrophils when responding to both artificial chemical activators and live pathogens.
“It was the first step of basically validating our technique to better understand neutrophils with known activators and techniques,” says Datta. “But I also wanted to see if the changes we were detecting with our optical method was also true in an in vivo model.”
They demonstrated using the technology within a living system by using zebrafish larvae. Zebrafish are a common animal model unique for their transparent appearance, allowing researchers to observe internal tissues and organs.
Datta says that zebrafish naturally have a lot of pigmentation, which interferes with the imaging. They used a special mutant strain of zebrafish called Casper, which results in a crystal-clear fish at all stages of development. Then to identify neutrophils, they used a special label called mCherry, a red fluorescent protein.
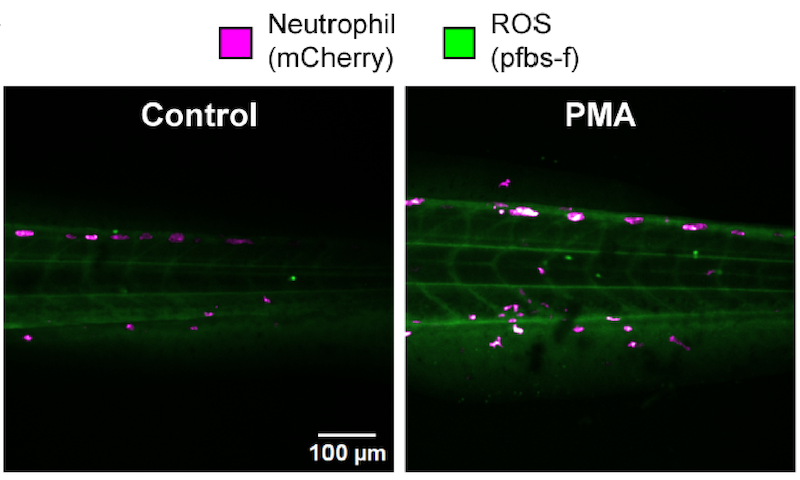
“We can see this beautiful tissue, and I can identify the borders of my neutrophils with the red label,” says Datta. “The in vivo imaging was really cool.”
This research provides evidence that OMI is uniquely suited for studying the fast-paced metabolism of neutrophils at the single-cell level and is useful in both cell culture and live models.
Datta says moving forward, they hope to continue collaborating with other labs to study neutrophils in the context of different diseases, such as cystic fibrosis or sepsis.
“Neutrophils have been understudied, and our specific technique is underused — most people don’t know about it yet,” she says. “Our lab loves collaborating. It’s really exciting to share our technology with so many different branches of research.”