Tag: cancer
At the Morgridge Institute, scientists across biology are working to better understand and stop cancer.
Megan Spurgeon: How a mild-mannered virus becomes a cancer culprit
Merkel cell carcinoma is the most recently discovered example of a cancer triggered by exposure to a virus. Morgridge Investigator Megan Spurgeon discusses how this rare and deadly form of skin cancer has become a major focal point of her research.
Cancer immunotherapy startup enters new chapter with SBIR support
Support from the National Science Foundation’s Small Business Innovation Research (SBIR) program is helping a Morgridge research team bring an important cancer immunotherapy technology to market.
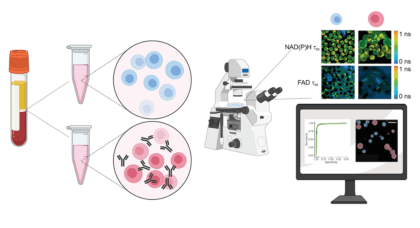
Researchers look to advanced metabolic imaging to improve cancer immunotherapy
Researchers at the Morgridge Institute and UW–Madison are using advanced metabolic imaging to identify ways to improve CAR T cell therapy against solid tumors.
Study shows environmental conditions alter dependence on a key metabolic enzyme
At the intersection of genetic rules and environmental conditions, scientists at Morgridge are gathering new insights about how diseases function in the human body.
Shaping the Future of Women’s Health: Insights and Innovations
Despite the many advances in human health, research focused on women’s health has a more complex history. Experts discussed this topic through the lens of discovery, prevention, and access to care during a Fearless Science Speaker Series lecture on March 17, 2025.
Morgridge postdoc breaks down barriers of cancer therapy at Falling Walls Lab Competition
Morgridge postdoc Amani Gillette took center stage at the international Falling Walls Lab Competition, presenting a pitch for SeLight, LLC, her startup focused on CAR T cell therapy research.
New Morgridge investigator explores complex link between viruses, cancer
Viruses are responsible for nearly one in every five cases of human cancer. New Morgridge Investigator Megan Spurgeon is on the front lines of understanding the inner workings of this public health threat.
Morgridge scientist aims to shatter the ‘ballistic barrier’ in imaging
With support from the Chan Zuckerberg Initiative (CZI), Morgridge investigator Randy Bartels will be on a quest to break the “ballistic barrier” in biomedical imaging to peer more deeply into living tissue.
Skala Lab’s photonics-based imaging featured in UW Health News
via UW Health
Melissa Skala’s technology harnesses the natural light produced by cells in order to track response to cancer drug treatment.
Juan Caicedo joins Morgridge, UW biomedical imaging community
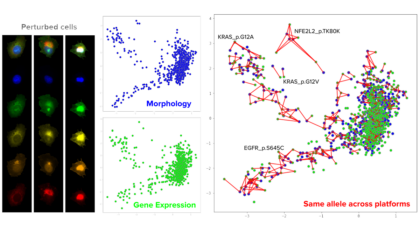
Meet new Morgridge biomedical imaging Investigator Juan Caicedo, who specializes in machine learning to decode complex patterns in human biology.
Visualizing metabolic activity of tumor and immune cells with melanoma-bearing mice
A study on metabolic reprogramming in cancer and immune cells is helping scientists better understand the cell interactions surrounding cancer progression — a key to developing more effective and personalized treatments.
CAR macrophages show promise for cancer immunotherapy treatments
Scientists use stem cells to develop CAR macrophages that can infiltrate and kill tumor cells, showing promise for cancer immunotherapy treatments.
Mass spec fueling new explorations at the Carbone Cancer Center
Every cell has about 20,000 proteins, several hundred metabolites, and couple of thousand lipids. When those things get perturbed, bad things can happen. But measuring those changes may offer essential clues for fighting cancer.
Metabolism research has morphed from mundane to essential
At a meeting of the Wisconsin Tech Council, Morgridge scientists described the renaissance taking place in metabolism research, which is revealing newfound connections to disease and entirely new roads to treatment and prevention.
Bernstein wraps postdoc, joins cancer research startup
After years of building tools for bioinformatics research, Morgridge Postdoctoral Fellow Matt Bernstein takes his skills to industrial cancer research.
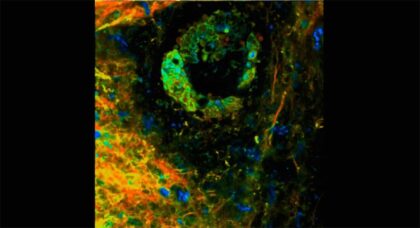
Optical imaging highlights metabolic interactions that make pancreatic tumor cells grow
An advanced biomedical imaging technique reveals how cancer cells can hijack the metabolic activity of certain non-cancer cells in the pancreas to fuel tumor growth.
Navigating the unCHARTed: web tool explores public sequencing data for cancer research
Morgridge Postdoctoral Fellow Matthew Bernstein developed a web tool to explore public RNAseq datasets to facilitate analysis for cancer researchers.
The (cell) medium is the message: A new tool for studying cell biology in a dish
Morgridge investigator Jason Cantor is partnering with Thermo Fisher Scientific to give biologists a new medium to study human cells in their most natural state.
Imaging technique sheds light on a notoriously slow-growing cancer
Neuroendocrine cancers grow so slowly they often evade detection before it’s too late. By mimicking that slow growth in the lab, the Melissa Skala Lab hopes to speed up the creation of more effective treatments.
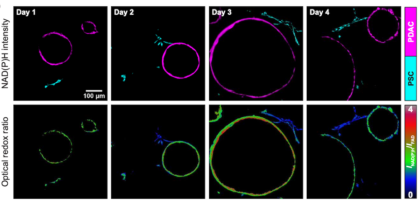
Redox imaging allows measurement of drug responses in lab-grown cancer samples
Scientists have developed a nondestructive way of measuring drug treatment responses in lab-grown cancer samples.
Researchers publish findings on use of a more human-like cell culture medium to explore gene essentiality
The Jason Cantor Lab at Morgridge is utilizing a new cell culture medium to ask how critical genes are to the survival and reproduction of human cells under different growth conditions. The technique could have important ramifications for the treatment of human diseases.
Breaking down barriers to pancreatic cancer care in Wisconsin
Researchers at the Morgridge Institute and UW–Madison want to ensure that pancreatic cancer treatment options are accessible to all — regardless of race, ethnicity, or insurance status — so that patients can make the most informed decisions regarding their care.
New technology tracks role of macrophages in cancer spread
A Morgridge imaging study of macrophages — immune cells that are important to human health, but paradoxically can help some cancers grow and spread — is offering better ways to understand these cells and target them with immunotherapies.
‘Like a Map of the City’ – How Metabolic Networks Contribute to Our Understanding of Cancer’s Growth
via UW Health
When Morgridge investigator Jing Fan thinks about metabolism, she is focused on the complicated network of biochemical reactions. Her lab has been working on understanding metabolism in a quantitative, systematic way.
Novel label-free imaging technique brings out the inner light within T cells
A new imaging method developed by the Skala lab uses the natural autofluorescence within cells to assess T cell activity. The technique could help assess T cell involvement in immunotherapies.
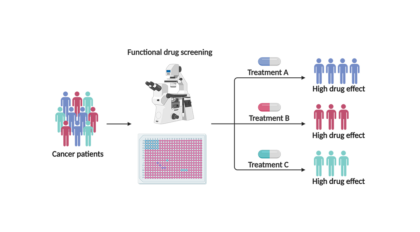
Improving the odds: Researchers develop novel device to study early stage of breast cancer
Imagine your chances of developing an invasive cancer were the same as a coin toss. Do you opt for aggressive treatments like surgery and chemotherapy, or do you take your chances that the cancer will never manifest?
Turning T cells into better cancer assassins
Alexandra Walsh, an assistant scientist in the lab of Morgridge medical engineer Melissa Skala, is leading a project to use non-invasive fluorescence imaging to identify and sort T cells for use in cancer immunotherapy treatments. The technology won a 2018 Innovation Award from the Wisconsin Alumni Research Foundation.
Pushing toward personalized pancreatic cancer treatments
Melissa Skala and Paul Campagnola, a professor of biomedical engineering at UW–Madison, hope to make inroads toward improved drug therapies through a two-year National Institutes of Health Exploratory/Developmental Research Grant.
Morgridge researcher scores in cancer research ‘lightning round’
Melissa Skala, a Morgridge Institute for Research investigator in medical engineering, won a highly competitive award from the nonprofit organization Stand Up to Cancer at its annual 2017 summit January in Santa Monica.
Getting personal with pancreatic cancer
Oncologists are struggling to improve the grim survival rates of pancreatic cancer, which are especially frustrating in an era that is making good progress on other cancer fronts. “I think
Melissa Skala: Follow the Light to Better Cancer Treatment
Skala’s research problems focus on cancer detection and treatment, and her expertise in light-based, optical imaging is giving clinicians revolutionary new tools for the fight. Skala will be bringing her talents this summer from Vanderbilt University to the Morgridge Institute for Research and the University of Wisconsin–Madison, as a Morgridge investigator and professor of Biomedical Engineering (BME).
Study redefines role of estrogen in cervical cancer
Scientists know that the hormone estrogen is a major driver in the growth of cervical cancer, but a new study examining genetic profiles of 128 clinical cases reached a surprising conclusion: Estrogen receptors all but vanish in cervical cancer tumors.
A pathway for understanding cancer’s origin
The tools of modern biology have made it possible to obtain an incredibly detailed picture of how cancer cells differ from healthy cells at the molecular level. Somewhat paradoxically, despite these meticulous portraits of cancer, it remains remarkably difficult to answer the very fundamental question: What caused cancer in this patient?